Every FDA-Approved Presbyopia Eye Drop Explained
Four prescription eye drops can now temporarily restore near vision without reading glasses. Here's what the clinical trials actually show — the good, the bad, and what your doctor might not mention.
Quick summary: All four drops work by constricting your pupil to create a "pinhole effect" that extends depth of focus. They differ in duration (6-10 hours), side effects, and which patients they work best for. Take our free candidacy quiz →
How Presbyopia Eye Drops Work
Presbyopia happens because the lens inside your eye gradually hardens with age, losing the flexibility needed to focus on near objects. Eye drops can't reverse this process — but they can work around it.
💡 The Pinhole Principle
All FDA-approved presbyopia drops work by constricting the pupil (miosis). A smaller pupil acts like a pinhole camera — it extends the depth of focus so that near objects appear sharper, even though the hardened lens hasn't changed. Think of it like squinting: the same principle, but pharmaceutical and more precise.
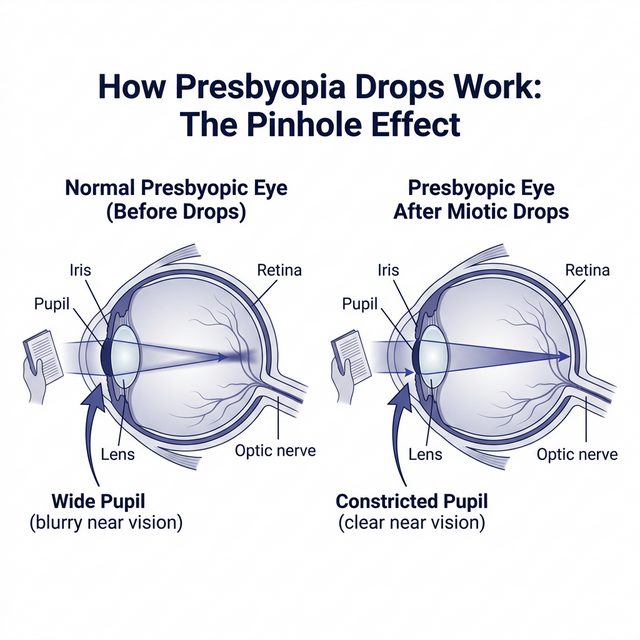
The pinhole effect: miotic drops constrict the pupil, increasing depth of focus
✓ What drops CAN do
- • Temporarily improve near vision for 6-10 hours
- • Allow reading without glasses in good lighting
- • Work within 15-30 minutes of application
- • Complement other treatments like brain training
✗ What drops CAN'T do
- • Reverse or cure presbyopia
- • Provide permanent improvement
- • Work well in dim lighting (constricted pupil)
- • Replace reading glasses for all situations
Head-to-Head Comparison

| Feature | Vuity | Qlosi | VIZZ | Yuvezzi |
|---|---|---|---|---|
| Active ingredient | Pilocarpine 1.25% | Pilocarpine 0.4% | Aceclidine 1.44% | Carbachol + Brimonidine |
| FDA Approved | Oct 2021 | Oct 2023 | Jul 2025 | Jan 2026 |
| Duration | ~6 hours | ~8 hours | ~10 hours | ~10 hours |
| Responder rate | ~31% | ~26% | ~71% | ~50% |
| Headache rate | ~15% | 5-7% | Low | Low |
| Dim vision | Significant | Moderate | Mild (pupil-selective) | Reduced |
| Post-LASIK safe | Caution | Caution | Yes | Not studied |
| Cost (approx.) | ~$80/mo | ~$75/mo | ~$85/mo | ~$100/mo |
| Detailed review | Read → | Read → | Read → | Read → |
Vuity (Pilocarpine 1.25%)
First to MarketThe first FDA-approved presbyopia drop (October 2021)1. Uses AbbVie's pHast technology for comfortable pH-adjusted delivery. Based on pilocarpine, a well-known muscarinic agonist used for decades in glaucoma treatment.
Real-world feedback often cites "dim vision" and headaches, especially in first weeks. Many users report diminishing efficacy over months. Best for: situational use in well-lit environments.
Read complete Vuity review →Qlosi (Pilocarpine 0.4%)
Low-Dose OptionApproved October 2023 by Orasis Pharmaceuticals. Uses a lower concentration of pilocarpine (0.4% vs Vuity's 1.25%) with a proprietary sustained-release formulation designed to reduce side effects while extending duration to ~8 hours.
Duration
~8 hours
Trial
26% gained 3+ lines
Advantage
Fewer headaches
The lower pilocarpine dose reduces headache frequency (5-7% vs 15%) and dim vision complaints. Tradeoff: slightly lower responder rate. Best for: patients who tried Vuity but couldn't tolerate the side effects.
Read complete Qlosi review →VIZZ (Aceclidine 1.44%)
Pupil-SelectiveApproved July 2025 by Lenz Therapeutics3. Uses aceclidine, a different muscarinic agonist with a key advantage: it's "pupil-selective" — it constricts the pupil without significantly affecting the ciliary muscle. This means less dim vision and fewer headaches than pilocarpine-based drops.
The CLARITY trial showed dramatically better results than first-generation drops. Aceclidine has been used in European ophthalmology since the 1960s. Particularly promising for post-LASIK plano-presbyopes who don't need distance correction.
Read complete VIZZ review →Yuvezzi (Carbachol + Brimonidine)
Newest (Jan 2026)Approved January 2026 by Ocuphire Pharma5. The first combination drop, pairing carbachol (a cholinergic for pupil constriction) with brimonidine (an alpha-agonist that counteracts the redness caused by miosis). A novel dual-mechanism approach.
Yuvezzi is notable for its extensive 12-month safety dataset (BRIO II trial) and its anti-redness benefit — brimonidine actually makes eyes look whiter rather than redder. Early days, but promising for patients who want longer-lasting effects with minimal cosmetic impact.
Read complete Yuvezzi review →⚠️ Safety: What Every Patient Should Know
All presbyopia eye drops share common safety considerations. For the complete deep-dive with clinical data, see our dedicated side effects guide.
Common Side Effects
- • Dim vision in low light — constricted pupil lets in less light
- • Headache — especially with pilocarpine-based drops, usually fades with use
- • Eye redness — less common with Yuvezzi (brimonidine counteracts this)
- • Blurred distance vision — tradeoff for better near vision
Serious Risks (Rare)
- • Retinal detachment — TriNetX cohort data7 shows elevated risk with pilocarpine. Higher risk in patients with high myopia or lattice degeneration
- • Pulfrich phenomenon — altered depth perception if applied to one eye
- • Chronic use concerns — potential for iris fibrosis, synechiae, cataractogenesis with long-term daily use
Who Should NOT Use Presbyopia Eye Drops
- • Patients with iritis or uveitis
- • People with high myopia (>-6D) due to retinal detachment risk
- • Night drivers (dim vision impairs driving safety)
- • Patients with lattice degeneration or retinal tears
The Smartest Approach: Drops + Brain Training
Eye drops fix the optics. Perceptual learning fixes the neural processing. Together, they address both sides of presbyopia.
💊 Eye Drops
- ✓ Instant effect (15-30 min)
- ✓ No training required
- ✗ Temporary (6-10 hours)
- ✗ Side effects possible
- ✗ Monthly cost ($75-100)
🧠 Gabor Patch Training
- ✓ Long-lasting improvements
- ✓ Zero side effects
- ✓ Improves neural processing
- ✗ Takes 2-4 weeks to see results
- ✗ Requires daily practice
Using both means you get the immediate relief of drops while building long-term neural improvements that may eventually reduce your dependence on drops.
Not Sure Which Treatment Is Right for You?
Our free assessment considers your age, vision status, lifestyle, and risk factors to give you a personalized recommendation.
People Also Ask
How many presbyopia eye drops are FDA approved?
As of early 2026, four presbyopia eye drops have received FDA approval: Vuity (pilocarpine 1.25%, approved October 2021), Qlosi (pilocarpine 0.4%, approved October 2023), VIZZ (aceclidine 1.44%, approved July 2025), and Yuvezzi (carbachol/brimonidine, approved January 2026). A fifth drop, CSF-1 by Orasis, is currently in Phase III trials.
Which presbyopia eye drop is the best?
There's no single "best" drop — it depends on your specific situation. VIZZ has the highest responder rate (71%) and longest duration (10 hours) with its pupil-selective mechanism. Yuvezzi offers anti-redness benefits. Qlosi is gentlest on headache-prone patients. Take our candidacy quiz or consult your eye doctor to find the best fit.
How much do presbyopia eye drops cost per month?
Presbyopia eye drops typically cost $75-100 per month without insurance. Most are not covered by standard vision or medical insurance because presbyopia is classified as a refractive condition rather than a disease. Some manufacturers offer savings programs. By comparison, Gabor patch brain training is a one-time investment with lasting results.
Are presbyopia eye drops safe to use long-term?
Short-term safety is well-established through FDA clinical trials. Long-term safety data is more limited since most trials lasted 3-12 months. Yuvezzi has the longest safety dataset (12 months in the BRIO II trial). Theoretical concerns about chronic miosis include iris fibrosis and posterior synechiae. Read our complete side effects guide for detailed safety information.
Sources
- U.S. Food and Drug Administration. VUITY (pilocarpine HCl ophthalmic solution) 1.25% Approval. October 2021.
- Price FW Jr, et al. "GEMINI 1 and GEMINI 2 Phase 3 Trials: Efficacy and Safety of AGN-190584 in Presbyopia." Ophthalmology. 2022;129(8):862-872.
- Lenz Therapeutics. FDA Approval of VIZZ (aceclidine ophthalmic solution) 1.44%. July 2025.
- Lenz Therapeutics. "CLARITY Phase 3 Results: Aceclidine 1.44% for Presbyopia." Ophthalmology. 2025.
- Ocuphire Pharma. FDA Approval of Yuvezzi (carbachol/brimonidine). January 2026.
- Ocuphire Pharma. "BRIO II: 12-Month Safety and Efficacy of Carbachol/Brimonidine for Presbyopia." Ophthalmology 360. 2025.
- American Academy of Ophthalmology. "Retinal Detachment Risk with Pilocarpine for Presbyopia: TriNetX Cohort Analysis." AAO Clinical Statement. 2024.
- American Society of Cataract and Refractive Surgery. "Consensus Statement on Presbyopia Drops: Pre-Screening Recommendations." 2024.